| << Chapter < Page | Chapter >> Page > |
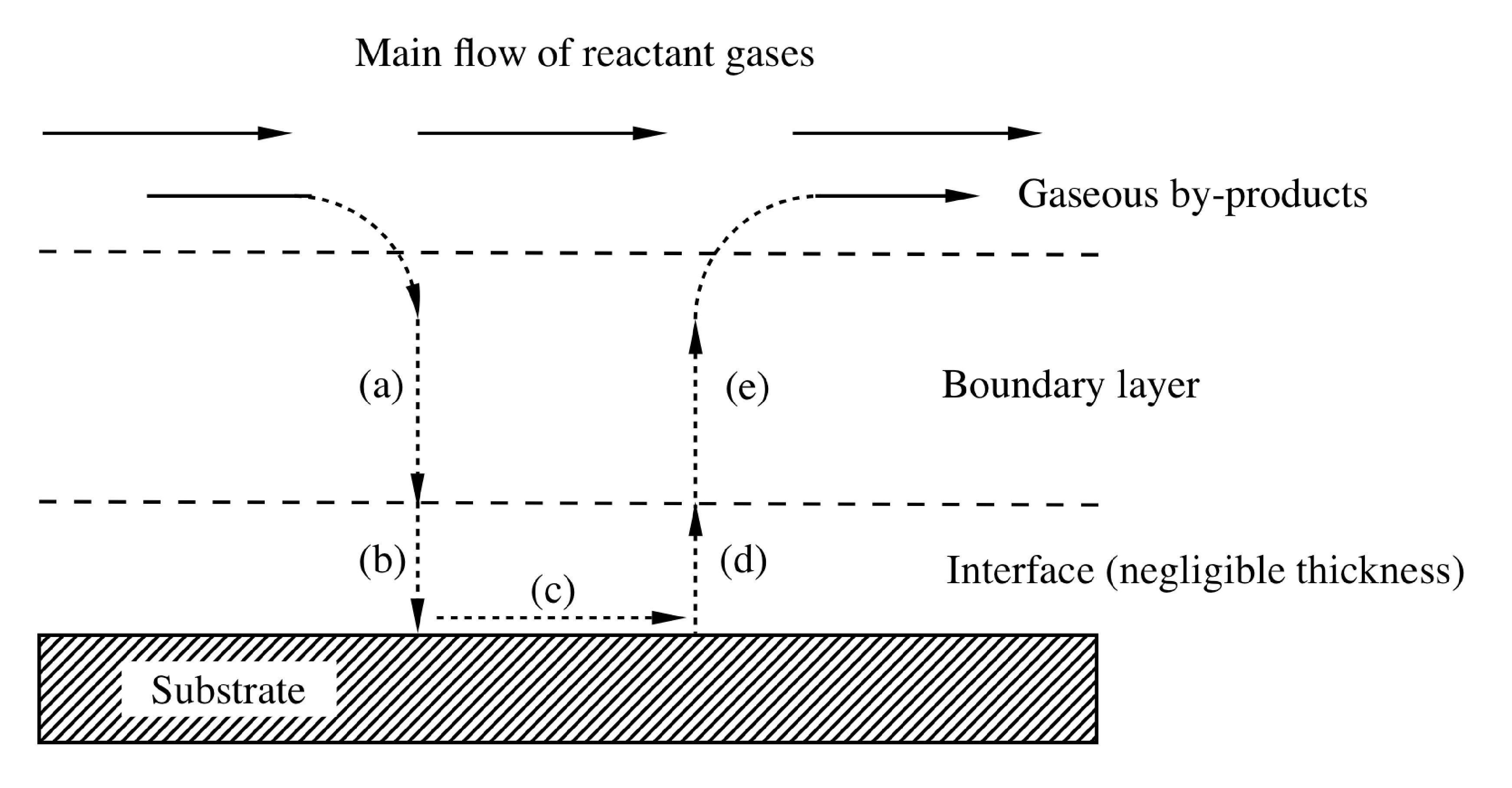
Gas flow in a CVD reactor is generally laminar, although in some cases heating of the chamber walls will create convection currents. The complete problem of gas flow through the system is too complex to be described here; however, assuming we have laminar flow (often a safe assumption) the gas velocity at the chamber walls will be zero. Between the wall (zero velocity) and the bulk gas velocity there is a boundary layer. The boundary layer thickness increases with lowered gas velocity and the distance from the tube inlet ( [link] ). Reactant gases flowing in the bulk must diffuse through the boundary layer to reach the substrate surface. Often, the susceptor is tilted to partially compensate for the increasing boundary-layer thickness and concentration profile.

During CVD the growth rate of the film is limited by either surface reaction kinetics, mass transport (diffusion) of precursors to the substrate, or the feed rate of the precursors.
Surface reaction controls the rate when growth occurs at low temperatures (where the reaction occurs slowly) and also dominates at low pressures (where the boundary layer is thin and reactants easily diffuse to the surface), see [link] . Since reactants easily diffuse through the boundary layer, the amount of reactant at the surface is independent of reactor pressure. Therefore, it is the reactions and motions of the precursors adsorbed on the surface which will determine the overall growth rate of the film. A sign of surface reaction limited growth would be dependence of the growth rate on substrate orientation, since the orientation would certainly not affect the thermodynamics or mass transport of the system.

A deposition limited by mass transport is controlled by the diffusion of reactants through the boundary layer and diffusion of by-products out of the boundary layer. Mass transport limits reactions when the temperature and pressure are high. These conditions increases the thickness of the boundary layer and make it harder for gases to diffuse through ( [link] ). In addition, decomposition of the reactants is typically quicker since the substrate is at a higher temperature. When mass transport limits the growth, either increasing the gas velocity or rotating the substrate during growth will decrease the boundary layer and increase the growth rate.

Notification Switch
Would you like to follow the 'Chemistry of electronic materials' conversation and receive update notifications?