| << Chapter < Page | Chapter >> Page > |
If the diode is reverse-biased, the terminal of the battery is connected to the n-type semiconductor. This makes it even more negatively charged. It also removes even more of the free electrons near the depletion band. At the same time, the terminal of the battery is connected to the p-type silicon. This will supply free electrons and fill in more of the holes next to the depletion band. Both processes cause the depletion band to get wider. The resistance of the diode (which was already high) increases. This is why a reverse-biased diode does not conduct.
Another explanation for the increased resistance is that the battery has made the p-type semiconductor m ore negative than it used to be, making it repel any electrons from the n-type semiconductor which attempt to cross the depletion band.
On the other hand, if the diode is forward biased, the depletion band is made narrower. The negative charge on the p-type silicon is cancelled out by the battery. The greater the voltage used, the narrower the depletion band becomes. Eventually, when the voltage is about 0,6 V (for silicon) the depletion band disappears. Once this has occurred, the diode conducts very well.
A light-emitting diode (LED) is a diode device that emits light when charge flows in the correct direction through it. If you apply a voltage to force current to flow in the direction the LED allows, it will light up.
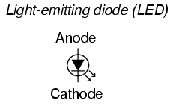
This notation of having two small arrows pointing away from the device is common to the schematic symbols of all light-emitting semiconductor devices. Conversely, if a device is light-activated (meaning that incoming light stimulates it), then the symbol will have two small arrows pointing toward it. It is interesting to note, though, that LEDs are capable of acting as light-sensing devices: they will generate a small voltage when exposed to light, much like a solar cell on a small scale. This property can be gainfully applied in a variety of light-sensing circuits.
The colour depends on the semiconducting material used to construct the LED, and can be in the near-ultraviolet, visible or infrared part of the electromagnetic spectrum.
Nick Holonyak Jr. (1928) of the University of Illinois at Urbana-Champaign developed the first practical visible-spectrum LED in 1962.
The wavelength of the light emitted, and therefore its colour, depends on the materials forming the p-n junction. A normal diode, typically made of silicon or germanium, emits invisible far-infrared light (so it can't be seen), but the materials used for an LED can emit light corresponding to near-infrared, visible or near-ultraviolet frequencies.

Notification Switch
Would you like to follow the 'Siyavula textbooks: grade 12 physical science' conversation and receive update notifications?