| << Chapter < Page | Chapter >> Page > |
When a dilute gas expands quasi-statically from 0.50 to 4.0 L, it does 250 J of work. Assuming that the gas temperature remains constant at 300 K, (a) what is the change in the internal energy of the gas? (b) How much heat is absorbed by the gas in this process?
In a quasi-static isobaric expansion, 500 J of work are done by the gas. The gas pressure is 0.80 atm and it was originally at 20.0 L. If the internal energy of the gas increased by 80 J in the expansion, how much heat does the gas absorb?
580 J
An ideal gas expands quasi-statically and isothermally from a state with pressure p and volume V to a state with volume 4V. How much heat is added to the expanding gas?
As shown below, if the heat absorbed by the gas along AB is 400 J, determine the quantities of heat absorbed along (a) ADB; (b) ACB; and (c) ADCB.
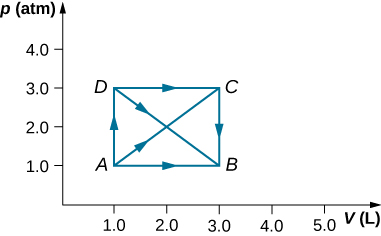
a. 600 J; b. 600 J; c. 800 J
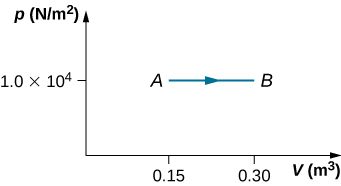
During the isobaric expansion from A to B represented below, 130 J of heat are removed from the gas. What is the change in its internal energy?
(a) What is the change in internal energy for the process represented by the closed path shown below? (b) How much heat is exchanged? (c) If the path is traversed in the opposite direction, how much heat is exchanged?
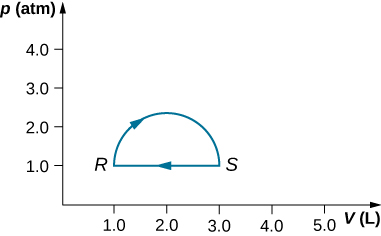
a. 0; b. 160 J; c. –160 J
When a gas expands along path AC shown below, it does 400 J of work and absorbs either 200 or 400 J of heat. (a) Suppose you are told that along path ABC, the gas absorbs either 200 or 400 J of heat. Which of these values is correct? (b) Give the correct answer from part (a), how much work is done by the gas along ABC? (c) Along CD, the internal energy of the gas decreases by 50 J. How much heat is exchanged by the gas along this path?
When a gas expands along AB (see below), it does 500 J of work and absorbs 250 J of heat. When the gas expands along AC , it does 700 J of work and absorbs 300 J of heat. (a) How much heat does the gas exchange along BC ? (b) When the gas makes the transmission from C to A along CDA , 800 J of work are done on it from C to D . How much heat does it exchange along CDA ?
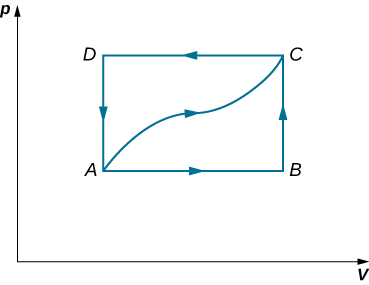
a. –150 J; b. –400 J
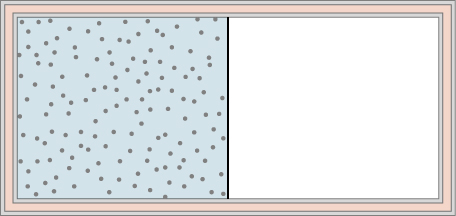
A dilute gas is stored in the left chamber of a container whose walls are perfectly insulating (see below), and the right chamber is evacuated. When the partition is removed, the gas expands and fills the entire container. Calculate the work done by the gas. Does the internal energy of the gas change in this process?
Ideal gases A and B are stored in the left and right chambers of an insulated container, as shown below. The partition is removed and the gases mix. Is any work done in this process? If the temperatures of A and B are initially equal, what happens to their common temperature after they are mixed?
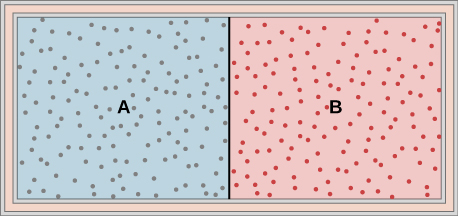
No work is done and they reach the same common temperature.
An ideal monatomic gas at a pressure of and a temperature of 300 K undergoes a quasi-static isobaric expansion from (a) What is the work done by the gas? (b) What is the temperature of the gas after the expansion? (c) How many moles of gas are there? (d) What is the change in internal energy of the gas? (e) How much heat is added to the gas?
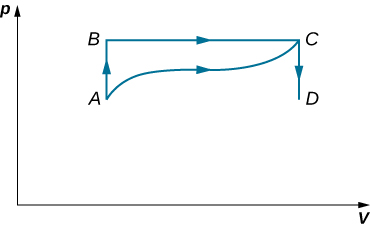
Consider the process for steam in a cylinder shown below. Suppose the change in the internal energy in this process is 30 kJ. Find the heat entering the system.
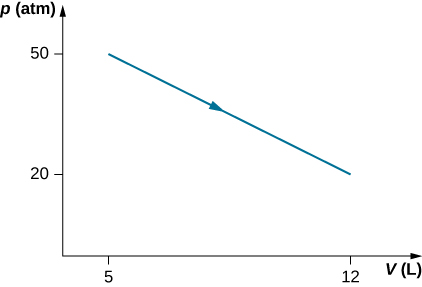
54,500 J
The state of 30 moles of steam in a cylinder is changed in a cyclic manner from a-b-c-a, where the pressure and volume of the states are: a (30 atm, 20 L), b (50 atm, 20 L), and c (50 atm, 45 L). Assume each change takes place along the line connecting the initial and final states in the pV plane. (a) Display the cycle in the pV plane. (b) Find the net work done by the steam in one cycle. (c) Find the net amount of heat flow in the steam over the course of one cycle.
A monatomic ideal gas undergoes a quasi-static process that is described by the function , where the starting state is and the final state . Assume the system consists of n moles of the gas in a container that can exchange heat with the environment and whose volume can change freely. (a) Evaluate the work done by the gas during the change in the state. (b) Find the change in internal energy of the gas. (c) Find the heat input to the gas during the change. (d) What are initial and final temperatures?
a. ; b. ; c. the sum of parts (a) and (b); d. and
A metallic container of fixed volume of immersed in a large tank of temperature contains two compartments separated by a freely movable wall. Initially, the wall is kept in place by a stopper so that there are 0.02 mol of the nitrogen gas on one side and 0.03 mol of the oxygen gas on the other side, each occupying half the volume. When the stopper is removed, the wall moves and comes to a final position. The movement of the wall is controlled so that the wall moves in infinitesimal quasi-static steps. (a) Find the final volumes of the two sides assuming the ideal gas behavior for the two gases. (b) How much work does each gas do on the other? (c) What is the change in the internal energy of each gas? (d) Find the amount of heat that enters or leaves each gas.
A gas in a cylindrical closed container is adiabatically and quasi-statically expanded from a state A (3 MPa, 2 L) to a state B with volume of 6 L along the path (a) Plot the path in the pV plane. (b) Find the amount of work done by the gas and the change in the internal energy of the gas during the process.
a.
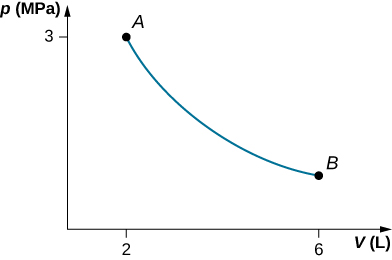 ;
;
b.

Notification Switch
Would you like to follow the 'University physics volume 2' conversation and receive update notifications?