| << Chapter < Page | Chapter >> Page > |
When comparing particle size data for the same sample represented using different distribution index, it is important to know that the results could be very different from number weighted distribution to intensity weighted distribution. This is clearly illustrated in the example below ( [link] ), for a sample consisting of equal numbers of particles with diameters of 5 nm and 50 nm. The number weighted distribution gives equal weighting to both types of particles, emphasizing the presence of the finer 5 nm particles, whereas the intensity weighted distribution has a signal one million times higher for the coarser 50 nm particles. The volume weighted distribution is intermediate between the two.
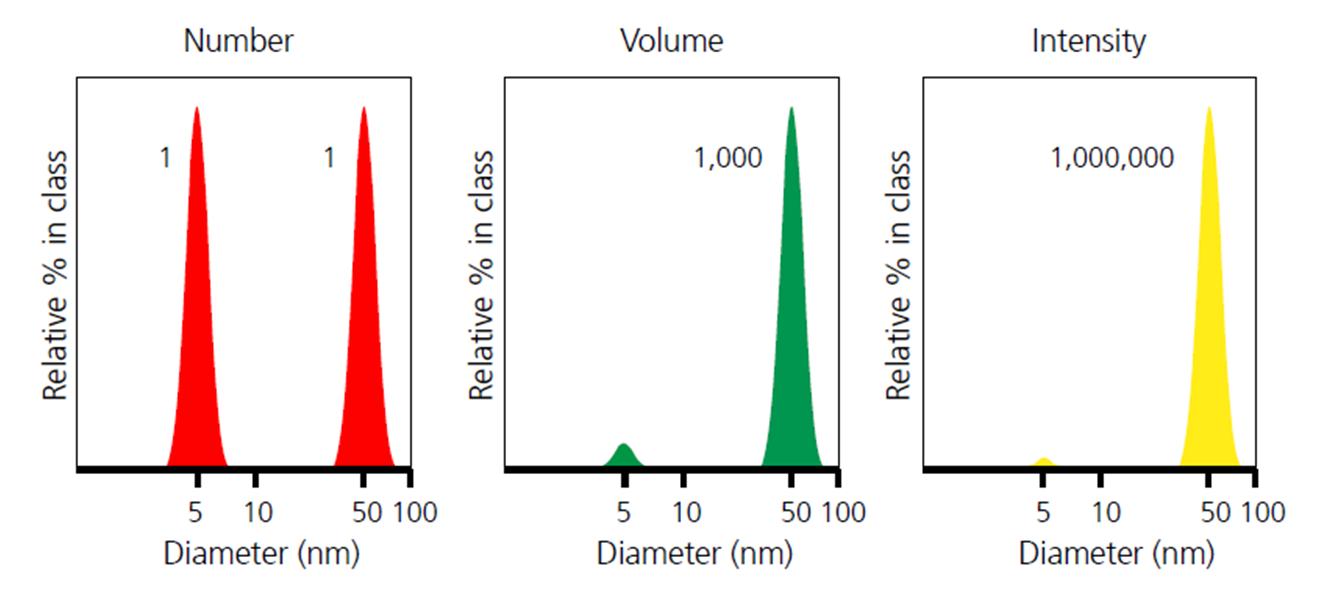
Furthermore, based on the different orders of correlation between the particle contribution and the particle size a , it is possible to convert particle size data from one type of distribution to another type of distribution, and that is also why the DLS software can also give size distributions in three different forms (number, volume, and intensity), where the first two kinds are actually deducted from the raw data of intensity weighted distribution.
As the DLS method could be used in many areas towards size distribution such as polymers, proteins, metal nanoparticles, or carbon nanomaterials, here gives an example about the application of DLS in size-controlled synthesis of monodisperse gold nanoparticles.
The size and size distribution of gold particles are controlled by subtle variation of the structure of the polymer, which is used to stabilize the gold nanoparticles during the reaction. These variations include monomer type, polymer molecular weight, end-group hydrophobicity, end-group denticity, and polymer concentration; a total number of 88 different trials have been conducted based on these variations. By using the DLS method, the authors are able to determine the gold particle size distribution for all these trials rather easily, and the correlation between polymer structure and particle size can also be plotted without further processing the data. Although other sizing techniques such as UV-V spectroscopy and TEM are also used in this paper, it is the DLS measurement that provides a much easier and reliable approach towards the size distribution analysis.
Since DLS is not the only method available to determine the size distribution of particles, it is also necessary to compare DLS with the other common-used general sizing techniques, especially TEM and AFM.
First of all, it has to be made clear that both TEM and AFM measure particles that are deposited on a substrate (Cu grid for TEM, mica for AFM), while DLS measures particles that are dispersed in a solution. In this way, DLS will be measuring the bulk phase properties and give a more comprehensive information about the size distribution of the sample. And for AFM or TEM, it is very common that a relatively small sampling area is analyzed, and the size distribution on the sampling area may not be the same as the size distribution of the original sample depending on how the particles are deposited.
On the other hand however, for DLS, the calculating process is highly dependent on the mathematical and physical assumptions and models, which is, monomodal distribution (cumulant method) and spherical shape for the particles, the results could be inaccurate when analyzing non-monomodal distributions or non-spherical particles. Yet, since the size determining process for AFM or TEM is nothing more than measuring the size from the image and then using the statistic, these two methods can provide much more reliable data when dealing with “irregular” samples.
Another important issue to consider is the time cost and complication of size measurement. Generally speaking, the DLS measurement should be a much easier technique, which requires less operation time and also cheaper equipment. And it could be really troublesome to analysis the size distribution data coming out from TEM or AFM images without specially programmed software.
In addition, there are some special issues to consider when choosing size analysis techniques. For example, if the originally sample is already on a substrate (synthesized by the CVD method), or the particles could not be stably dispersed within solution, apparently the DLS method is not suitable. Also, when the particles tend to have a similar imaging contrast against the substrate (carbon nanomaterials on TEM grid), or tend to self-assemble and aggregate on the surface of the substrate, the DLS approach might be a better choice.
In general research work however, the best way to do size distribution analysis is to combine these analyzing methods, and get complimentary information from different aspects. One thing to keep in mind, since the DLS actually measures the hydrodynamic radius of the particles, the size from DLS measurement is always larger than the size from AFM or TEM measurement. As a conclusion, the comparison between DLS and AFM/TEM is shown in [link] .
| DLS | AFM/TEM | |
| Sample preparation | Solution | Substrate |
| Measurement | Easy | Difficult |
| Sampling | Bulk | Small area |
| Shape of particles | Sphere | No requirement |
| Polydispersity | Low | No requirement |
| Size range | nm to um | nm to um |
| Size info. | Hydrodynamic radius | Physical size |
In general, relying on the fluctuating Rayleigh scattering of small particles that randomly moves in solution, DLS is a very useful and rapid technique used in the size distribution of particles in the fields of physics, chemistry, and bio-chemistry, especially for monomodally dispersed spherical particles, and by combining with other techniques such as AFM and TEM, a comprehensive understanding of the size distribution of the analyte can be readily acquired.

Notification Switch
Would you like to follow the 'Nanomaterials and nanotechnology' conversation and receive update notifications?