| << Chapter < Page | Chapter >> Page > |
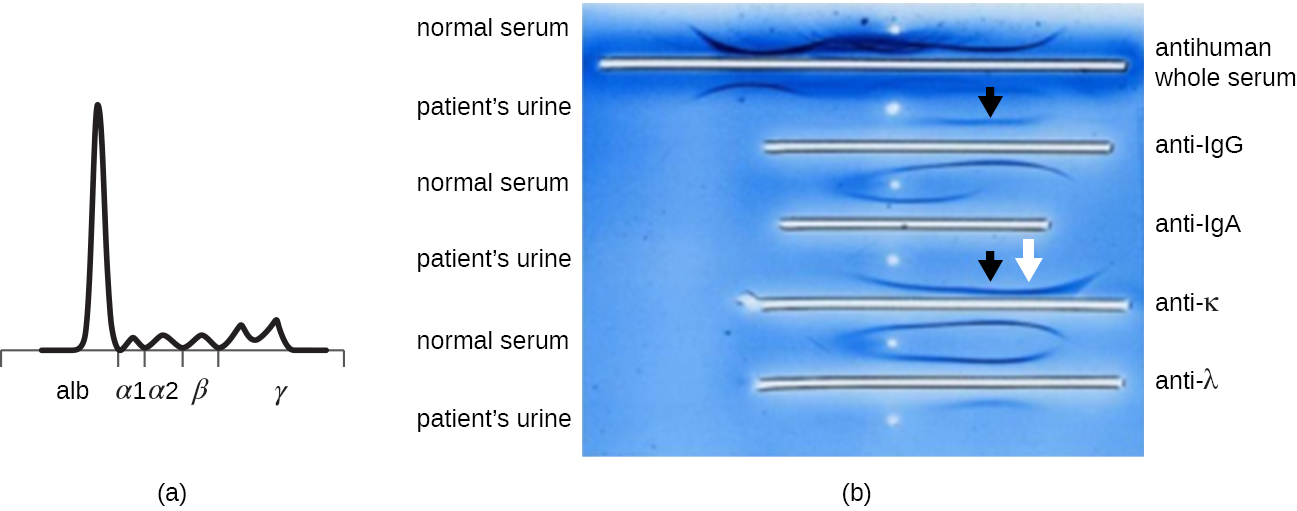
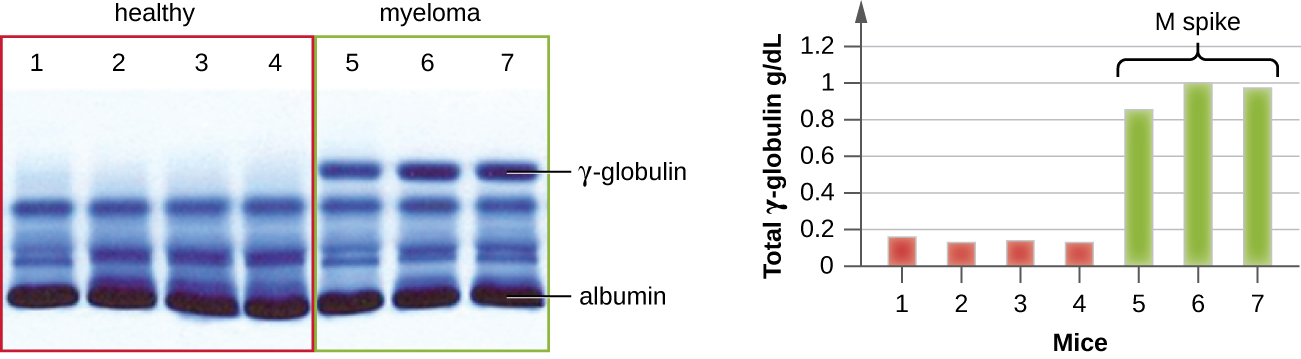
The advent of electrophoresis ultimately led to researching and understanding the structure of antibodies. When Swedish biochemist Arne Tiselius (1902–1971) published the first protein electrophoresis results in 1937, Tiselius, Arne, “Electrophoresis of Serum Globulin: Electrophoretic Analysis of Normal and Immune Sera,” Biochemical Journal 31, no. 9 (1937): 1464. he could identify the protein albumin (the smallest and most abundant serum protein) by the sharp band it produced in the gel. The other serum proteins could not be resolved in a simple protein electrophoresis, so he named the three broad bands, with many proteins in each band, alpha, beta, and gamma globulins. Two years later, American immunologist Elvin Kabat (1914–2000) traveled to Sweden to work with Tiselius using this new technique and showed that antibodies migrated as gamma globulins. Tiselius, Arne and Elvin A. Kabat. “An Electrophoretic Study of Immune Sera and Purified Antibody Preparations,” The Journal of Experimental Medicine 69, no. 1 (1939): 119-31. With this new understanding in hand, researchers soon learned that multiple myeloma, because it is a cancer of antibody-secreting cells, could be tentatively diagnosed by the presence of a large M spike in the gamma-globulin region by protein electrophoresis. Prior to this discovery, studies on immunoglobulin structure had been minimal, because of the difficulty of obtaining pure samples to study. Sera from multiple myeloma patients proved to be an excellent source of highly enriched monoclonal immunoglobulin, providing the raw material for studies over the next 20-plus years that resulted in the elucidation of the structure of immunoglobulin.
After performing protein gel electrophoresis, specific proteins can be identified in the gel using antibodies. This technique is known as the western blot . Following separation of proteins by PAGE, the protein antigens in the gel are transferred to and immobilized on a nitrocellulose membrane. This membrane can then be exposed to a primary antibody produced to specifically bind to the protein of interest. A second antibody equipped with a molecular beacon will then bind to the first. These secondary antibodies are coupled to another molecule such as an enzyme or a fluorophore (a molecule that fluoresces when excited by light). When using antibodies coupled to enzymes, a chromogenic substrate for the enzyme is added. This substrate is usually colorless but will develop color in the presence of the antibody. The fluorescence or substrate coloring identifies the location of the specific protein in the membrane to which the antibodies are bound ( [link] ).

Notification Switch
Would you like to follow the 'Microbiology' conversation and receive update notifications?