| << Chapter < Page | Chapter >> Page > |
Some gold sols prepared in 1857 are still intact (the particles have not coalesced and settled), illustrating the long-term stability of many colloids.
Pioneers made soap by boiling fats with a strongly basic solution made by leaching potassium carbonate, K 2 CO 3 , from wood ashes with hot water. Animal fats contain polyesters of fatty acids (long-chain carboxylic acids). When animal fats are treated with a base like potassium carbonate or sodium hydroxide, glycerol and salts of fatty acids such as palmitic, oleic, and stearic acid are formed. The salts of fatty acids are called soaps . The sodium salt of stearic acid, sodium stearate, has the formula C 17 H 35 CO 2 Na and contains an uncharged nonpolar hydrocarbon chain, the C 17 H 35 — unit, and an ionic carboxylate group, the — unit ( [link] ).
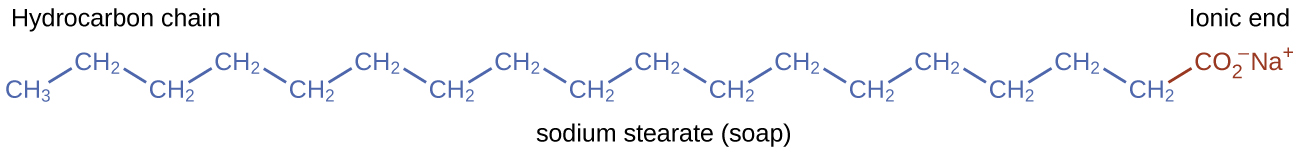
Detergents (soap substitutes) also contain nonpolar hydrocarbon chains, such as C 12 H 25 —, and an ionic group, such as a sulfate— or a sulfonate— ( [link] ). Soaps form insoluble calcium and magnesium compounds in hard water; detergents form water-soluble products—a definite advantage for detergents.

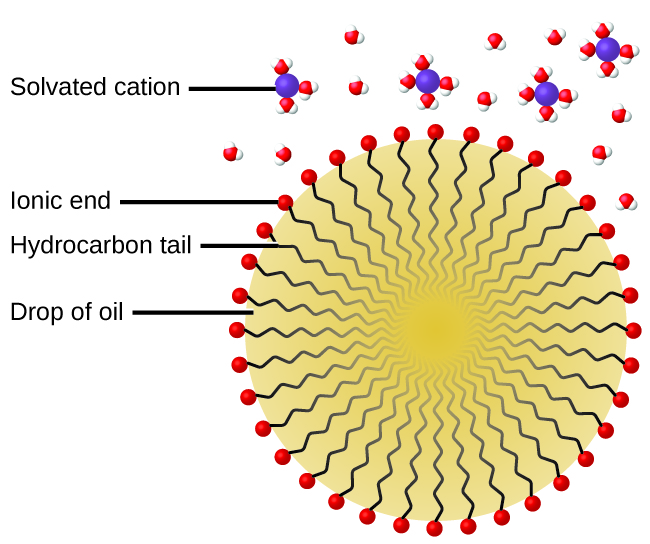
The cleaning action of soaps and detergents can be explained in terms of the structures of the molecules involved. The hydrocarbon (nonpolar) end of a soap or detergent molecule dissolves in, or is attracted to, nonpolar substances such as oil, grease, or dirt particles. The ionic end is attracted by water (polar), illustrated in [link] . As a result, the soap or detergent molecules become oriented at the interface between the dirt particles and the water so they act as a kind of bridge between two different kinds of matter, nonpolar and polar. Molecules such as this are termed amphiphilic since they have both a hydrophobic (“water-fearing”) part and a hydrophilic (“water-loving”) part. As a consequence, dirt particles become suspended as colloidal particles and are readily washed away.
The blowout of the Deepwater Horizon oil drilling rig on April 20, 2010, in the Gulf of Mexico near Mississippi began the largest marine oil spill in the history of the petroleum. In the 87 days following the blowout, an estimated 4.9 million barrels (210 million gallons) of oil flowed from the ruptured well 5000 feet below the water’s surface. The well was finally declared sealed on September 19, 2010.
Crude oil is immiscible with and less dense than water, so the spilled oil rose to the surface of the water. Floating booms, skimmer ships, and controlled burns were used to remove oil from the water’s surface in an attempt to protect beaches and wetlands along the Gulf coast. In addition to removal of the oil, attempts were also made to lessen its environmental impact by rendering it “soluble” (in the loose sense of the term) and thus allowing it to be diluted to hopefully less harmful levels by the vast volume of ocean water. This approach used 1.84 million gallons of the oil dispersant Corexit 9527, most of which was injected underwater at the site of the leak, with small amounts being sprayed on top of the spill. Corexit 9527 contains 2-butoxyethanol (C 6 H 14 O 2 ), an amphiphilic molecule whose polar and nonpolar ends are useful for emulsifying oil into small droplets, increasing the surface area of the oil and making it more available to marine bacteria for digestion ( [link] ). While this approach avoids many of the immediate hazards that bulk oil poses to marine and coastal ecosystems, it introduces the possibility of long-term effects resulting from the introduction of the complex and potential toxic components of petroleum into the ocean’s food chain. A number of organizations are involved in monitoring the extended impact of this oil spill, including the National Oceanic and Atmospheric Administration (visit this website for additional details).


Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?