| << Chapter < Page | Chapter >> Page > |
In [Fe(CN) 6 ] 4− , the strong field of six cyanide ligands produces a large Δ oct . Under these conditions, the electrons require less energy to pair than they require to be excited to the e g orbitals (Δ oct >P). The six 3 d electrons of the Fe 2+ ion pair in the three t 2 g orbitals ( [link] ). Complexes in which the electrons are paired because of the large crystal field splitting are called low-spin complexes because the number of unpaired electrons (spins) is minimized.
![A diagram is shown with a vertical arrow pointing upward along the height of the diagram at its left side. This arrow is labeled, “E.” to the right of this arrow are several rows of squares outlined in yellow. The first row has three linked squares at the lower right side of the diagram labeled, “[ F e ( C N ) subscript 6 ] superscript 4 negative sign.” Each square contains two half arrows, with the left half arrow pointing up and the right half arrow pointing down. A second row is positioned just above and to the left of the first, with the bottom of the squares at a height equal with the top of the squares in the first row. This group is also composed of 3 linked squares. The square to the left contains two half arrows, with the left half arrow pointing up and the right half arrow pointing down. The remaining two squares in this row only have upward pointing half arrows. At the bottom of the figure, just below this group is the label “[ F e ( H subscript 2 O ) subscript 6] superscript 2 plus sign.” To the left of this row, a third row that includes 5 linked squares is positioned with the lower edge of the squares at a height equal to the tops of the squares in row two. The square to the left contains two half arrows, one up and one down. All other squares in this group contain single upward pointing arrows. At the bottom of the figure beneath this row appears the label, “F e superscript 2 plus sign no ligands.” A fourth row composed of 2 linked squares appears to the right and above the second row. The lower edge of the squares is at a height level with the top of the squares of the third row. Each of these squares contains a single upward pointing half arrow. A fifth row of 2 squares is positioned above and to the right directly above the first row with the base of the squares positioned at a level equal to the top of the previous row of squares. These squares are empty. At the right of the diagram, a short horizontal line segment is drawn just right of the lower side of the rightmost square of the first row. A double-headed arrow extends from this line segment to a second horizontal line segment directly above the first and right of the lower side of the fifth row of squares. The arrow is labeled, “Low spin, large delta subscript oct,” to the right. The lower horizontal line segment is similarly labeled, “t subscript 2 g,” and the upper line segment is labeled, “e subscript g.” To the right of the second and fourth rows, a short horizontal line segment is drawn just right of the lower side of the rightmost square of the second row. A double-headed arrow extends from this line segment to a second horizontal line segment directly above the first and right of the lower side of the fourth row of squares. The arrow is labeled, “High spin, small capital delta subscript oct,” to the right. The lower horizontal line segment is similarly labeled, “t subscript 2 g,” and the upper line segment is labeled, “e subscript g.”](/ocw/mirror/col11760/m51188/CNX_Chem_19_03_Lowhigh.jpg)
In [Fe(H 2 O) 6 ] 2+ , on the other hand, the weak field of the water molecules produces only a small crystal field splitting (Δ oct <P). Because it requires less energy for the electrons to occupy the e g orbitals than to pair together, there will be an electron in each of the five 3 d orbitals before pairing occurs. For the six d electrons on the iron(II) center in [Fe(H 2 O) 6 ] 2+ , there will be one pair of electrons and four unpaired electrons ( [link] ). Complexes such as the [Fe(H 2 O) 6 ] 2+ ion, in which the electrons are unpaired because the crystal field splitting is not large enough to cause them to pair, are called high-spin complexes because the number of unpaired electrons (spins) is maximized.
A similar line of reasoning shows why the [Fe(CN) 6 ] 3− ion is a low-spin complex with only one unpaired electron, whereas both the [Fe(H 2 O) 6 ] 3+ and [FeF 6 ] 3− ions are high-spin complexes with five unpaired electrons.
(a) K 3 [CrI 6 ]
(b) [Cu(en) 2 (H 2 O) 2 ]Cl 2
(c) Na 3 [Co(NO 2 ) 6 ]
(a) Cr 3+ has a d 3 configuration. These electrons will all be unpaired.
(b) Cu 2+ is d 9 , so there will be one unpaired electron.
(c) Co 3+ has d 6 valence electrons, so the crystal field splitting will determine how many are paired. Nitrite is a strong-field ligand, so the complex will be low spin. Six electrons will go in the t 2 g orbitals, leaving 0 unpaired.
d 4 , d 5 , d 6 , and d 7
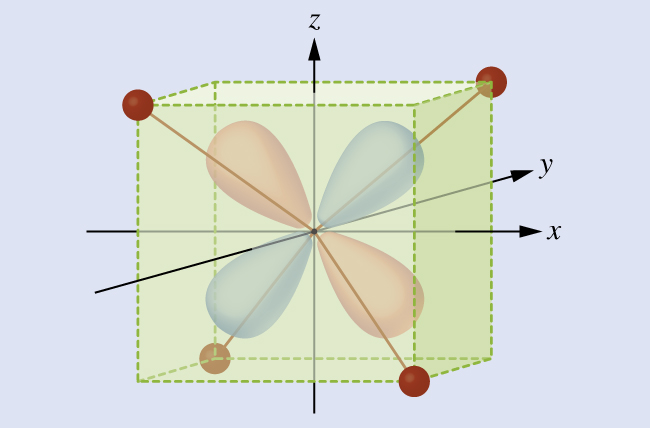
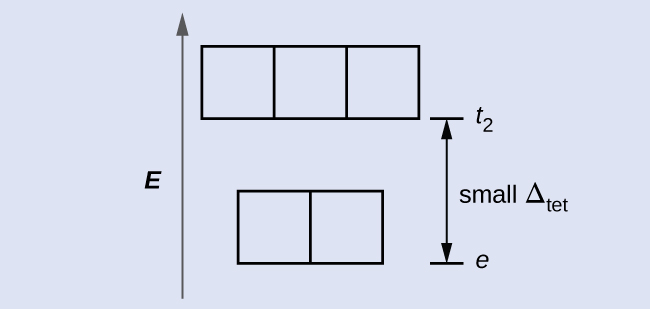
4; because Δ tet is small, all tetrahedral complexes are high spin and the electrons go into the t 2 orbitals before pairing

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?