| << Chapter < Page | Chapter >> Page > |
Reduction of boric oxide with magnesium powder forms boron (95–98.5% pure) as a brown, amorphous powder:
An amorphous substance is a material that appears to be a solid, but does not have a long-range order like a true solid. Treatment with hydrochloric acid removes the magnesium oxide. Further purification of the boron begins with conversion of the impure boron into boron trichloride. The next step is to heat a mixture of boron trichloride and hydrogen:
Silicon makes up nearly one-fourth of the mass of the earth’s crust—second in abundance only to oxygen. The crust is composed almost entirely of minerals in which the silicon atoms are at the center of the silicon-oxygen tetrahedron, which connect in a variety of ways to produce, among other things, chains, layers, and three-dimensional frameworks. These minerals constitute the bulk of most common rocks, soil, and clays. In addition, materials such as bricks, ceramics, and glasses contain silicon compounds.
It is possible to produce silicon by the high-temperature reduction of silicon dioxide with strong reducing agents, such as carbon and magnesium:
Extremely pure silicon is necessary for the manufacture of semiconductor electronic devices. This process begins with the conversion of impure silicon into silicon tetrahalides, or silane (SiH 4 ), followed by decomposition at high temperatures. Zone refining, illustrated in [link] , completes the purification. In this method, a rod of silicon is heated at one end by a heat source that produces a thin cross-section of molten silicon. Slowly lowering the rod through the heat source moves the molten zone from one end of the rod to other. As this thin, molten region moves, impurities in the silicon dissolve in the liquid silicon and move with the molten region. Ultimately, the impurities move to one end of the rod, which is then cut off.
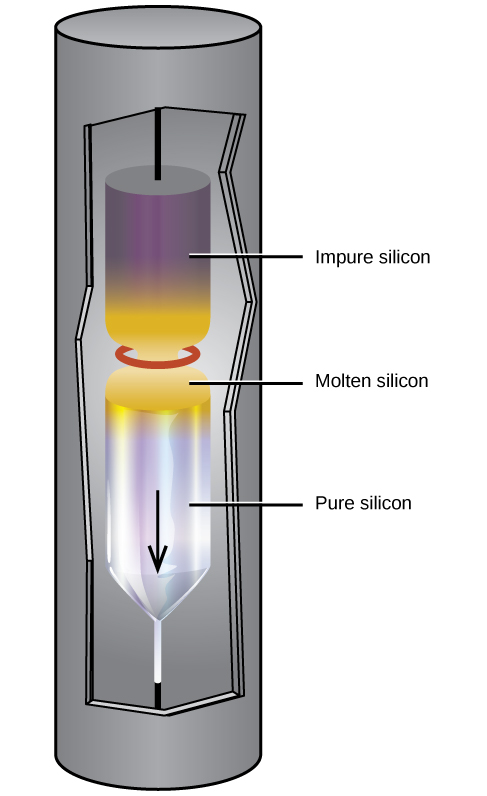
This highly purified silicon, containing no more than one part impurity per million parts of silicon, is the most important element in the computer industry. Pure silicon is necessary in semiconductor electronic devices such as transistors, computer chips, and solar cells.
Like some metals, passivation of silicon occurs due the formation of a very thin film of oxide (primarily silicon dioxide, SiO 2 ). Silicon dioxide is soluble in hot aqueous base; thus, strong bases destroy the passivation. Removal of the passivation layer allows the base to dissolve the silicon, forming hydrogen gas and silicate anions. For example:
Silicon reacts with halogens at high temperatures, forming volatile tetrahalides, such as SiF 4 .
Unlike carbon, silicon does not readily form double or triple bonds. Silicon compounds of the general formula SiX 4 , where X is a highly electronegative group, can act as Lewis acids to form six-coordinate silicon. For example, silicon tetrafluoride, SiF 4 , reacts with sodium fluoride to yield Na 2 [SiF 6 ], which contains the octahedral ion in which silicon is sp 3 d 2 hybridized:

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?