-
Home
- Chemistry
- Equilibria of other reaction
- Lewis acids and bases
Dissociation of a complex ion
Calculate the concentration of the silver ion in a solution that initially is 0.10
M with respect to
Solution
We use the familiar path to solve this problem:
-
Determine the direction of change. The complex ion
is in equilibrium with its components, as represented by the equation:
We write the equilibrium as a formation reaction because
Appendix K lists formation constants for complex ions. Before equilibrium, the reaction quotient is larger than the equilibrium constant [
K
f = 1.7
10
7 , and
it is infinitely large], so the reaction shifts to the left to reach equilibrium.
-
Determine x
and equilibrium concentrations. We let the change in concentration of Ag
+ be
x . Dissociation of 1 mol of
gives 1 mol of Ag
+ and 2 mol of NH
3 , so the change in [NH
3 ] is 2
x and that of
is –
x . In summary:
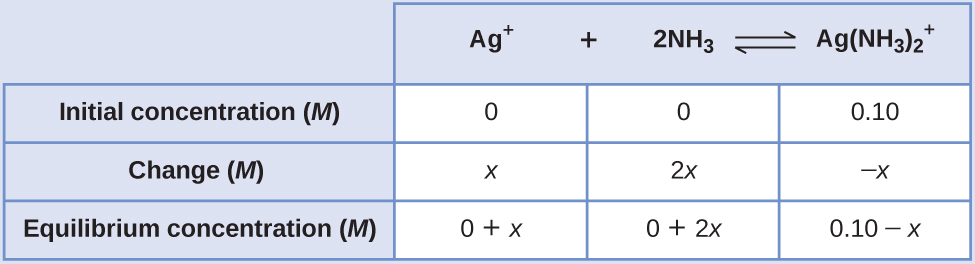
-
Solve for x and the equilibrium concentrations. At equilibrium:
Both
Q and
K
f are much larger than 1, so let us assume that the changes in concentrations needed to reach equilibrium are small. Thus 0.10 –
x is approximated as 0.10:
Because only 1.1% of the
dissociates into Ag
+ and NH
3 , the assumption that
x is small is justified.
Now we determine the equilibrium concentrations:
The concentration of free silver ion in the solution is 0.0011
M .
-
Check the work. The value of
Q calculated using the equilibrium concentrations is equal to
K
f within the error associated with the significant figures in the calculation.
Check your learning
Calculate the silver ion concentration, [Ag
+ ], of a solution prepared by dissolving 1.00 g of AgNO
3 and 10.0 g of KCN in sufficient water to make 1.00 L of solution. (Hint: Because
Q <
K
f , assume the reaction goes to completion then calculate the [Ag
+ ] produced by dissociation of the complex.)
Got questions? Get instant answers now!
Key concepts and summary
G.N. Lewis proposed a definition for acids and bases that relies on an atom’s or molecule’s ability to accept or donate electron pairs. A Lewis acid is a species that can accept an electron pair, whereas a Lewis base has an electron pair available for donation to a Lewis acid. Complex ions are examples of Lewis acid-base adducts. In a complex ion, we have a central atom, often consisting of a transition metal cation, which acts as a Lewis acid, and several neutral molecules or ions surrounding them called ligands that act as Lewis bases. Complex ions form by sharing electron pairs to form coordinate covalent bonds. The equilibrium reaction that occurs when forming a complex ion has an equilibrium constant associated with it called a formation constant,
K
f . This is often referred to as a stability constant, as it represents the stability of the complex ion. Formation of complex ions in solution can have a profound effect on the solubility of a transition metal compound.
Questions & Answers
number of sport play by 50 student construct discrete data
width of the frangebany leaves on how to write a introduction
Solve the mean of variance
Step 1: Find the mean. To find the mean, add up all the scores, then divide them by the number of scores. ...
Step 2: Find each score's deviation from the mean. ...
Step 3: Square each deviation from the mean. ...
Step 4: Find the sum of squares. ...
Step 5: Divide the sum of squares by n – 1 or N.
kenneth
Is mistake done to something
Vutshila
What is the life teble
anas
statistics is the analyzing of data
how do you calculate mean
diveving the sum if all values
Shaynaynay
let A1,A2 and A3 events be independent,show that (A1)^c, (A2)^c and (A3)^c are independent?
data collected all over the world
Shaynaynay
construct a less than and more than table
The sample of 16 students is taken. The average age in the sample was 22 years with astandard deviation of 6 years. Construct a 95% confidence interval for the age of the population.
Bhartdarshan' is an internet-based travel agency wherein customer can see videos of the cities they plant to visit. The number of hits daily is a normally distributed random variable with a mean of 10,000 and a standard deviation of 2,400
a. what is the probability of getting more than 12,000 hits?
b. what is the probability of getting fewer than 9,000 hits?
Bhartdarshan'is an internet-based travel agency wherein customer can see videos of the cities they plan to visit. The number of hits daily is a normally distributed random variable with a mean of 10,000 and a standard deviation of 2,400.
a. What is the probability of getting more than 12,000 hits
Akshay
Sorry i want to learn more about this question
Bright
a= 0.20233
b=0.3384
Sufiyan
How do I interpret level of significance?
It depends on your business problem or in Machine Learning you could use ROC- AUC cruve to decide the threshold value
Shivam
how skewness and kurtosis are used in statistics
Got questions? Join the online conversation and get instant answers!
Source:
OpenStax, Chemistry. OpenStax CNX. May 20, 2015 Download for free at http://legacy.cnx.org/content/col11760/1.9
Google Play and the Google Play logo are trademarks of Google Inc.


