| << Chapter < Page | Chapter >> Page > |
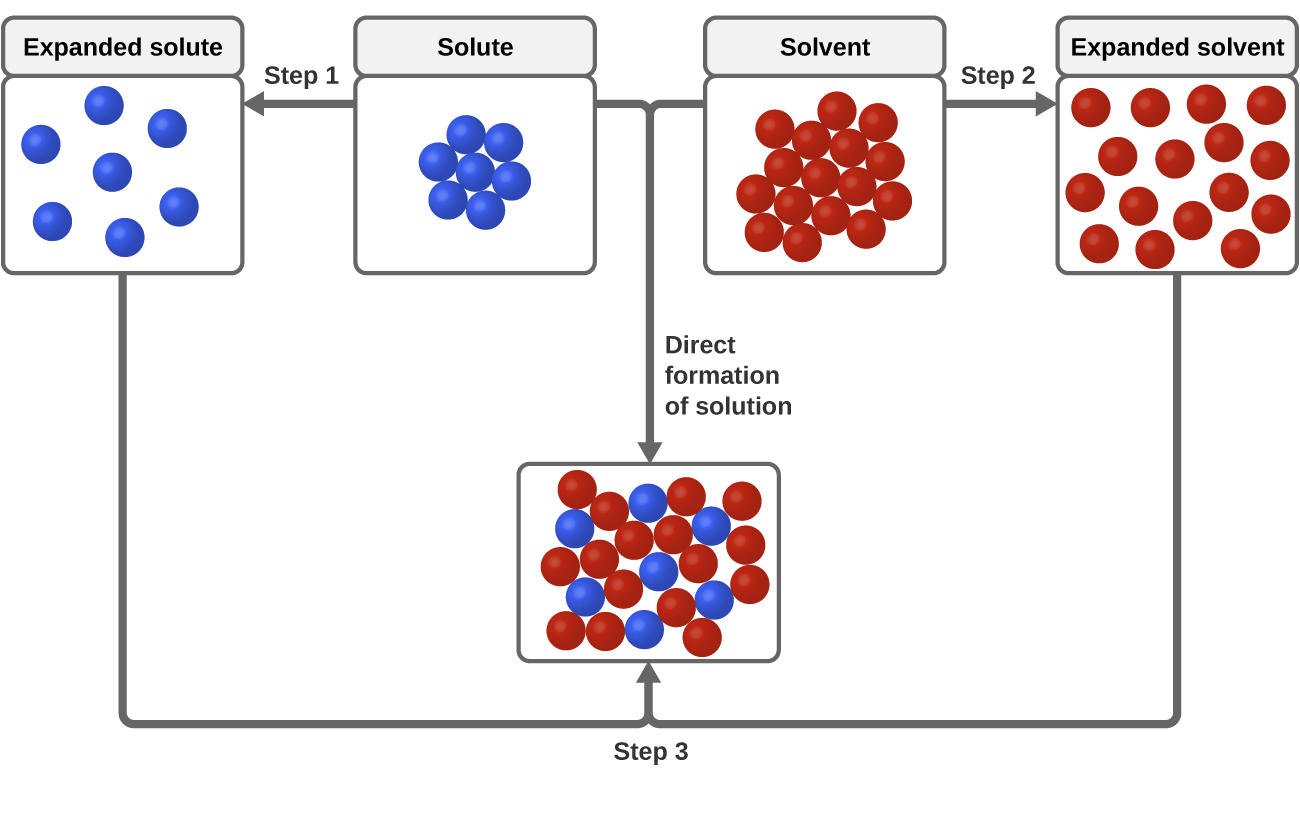
Three types of intermolecular attractive forces are relevant to the dissolution process: solute-solute, solvent-solvent, and solute-solvent. As illustrated in [link] , the formation of a solution may be viewed as a stepwise process in which energy is consumed to overcome solute-solute and solvent-solvent attractions (endothermic processes) and released when solute-solvent attractions are established (an exothermic process referred to as solvation ). The relative magnitudes of the energy changes associated with these stepwise processes determine whether the dissolution process overall will release or absorb energy. In some cases, solutions do not form because the energy required to separate solute and solvent species is so much greater than the energy released by solvation.
For example, cooking oils and water will not mix to any appreciable extent to yield solutions ( [link] ). Hydrogen bonding is the dominant intermolecular attractive force present in liquid water; the nonpolar hydrocarbon molecules of cooking oils are not capable of hydrogen bonding, instead being held together by dispersion forces. Forming an oil-water solution would require overcoming the very strong hydrogen bonding in water, as well as the significantly strong dispersion forces between the relatively large oil molecules. And, since the polar water molecules and nonpolar oil molecules would not experience very strong intermolecular attraction, very little energy would be released by solvation.

On the other hand, a mixture of ethanol and water will mix in any proportions to yield a solution. In this case, both substances are capable of hydrogen bonding, and so the solvation process is sufficiently exothermic to compensate for the endothermic separations of solute and solvent molecules.
As noted at the beginning of this module, spontaneous solution formation is favored, but not guaranteed, by exothermic dissolution processes. While many soluble compounds do, indeed, dissolve with the release of heat, some dissolve endothermically. Ammonium nitrate (NH 4 NO 3 ) is one such example and is used to make instant cold packs for treating injuries like the one pictured in [link] . A thin-walled plastic bag of water is sealed inside a larger bag with solid NH 4 NO 3 . When the smaller bag is broken, a solution of NH 4 NO 3 forms, absorbing heat from the surroundings (the injured area to which the pack is applied) and providing a cold compress that decreases swelling. Endothermic dissolutions such as this one require a greater energy input to separate the solute species than is recovered when the solutes are solvated, but they are spontaneous nonetheless due to the increase in disorder that accompanies formation of the solution.


Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?