| << Chapter < Page | Chapter >> Page > |
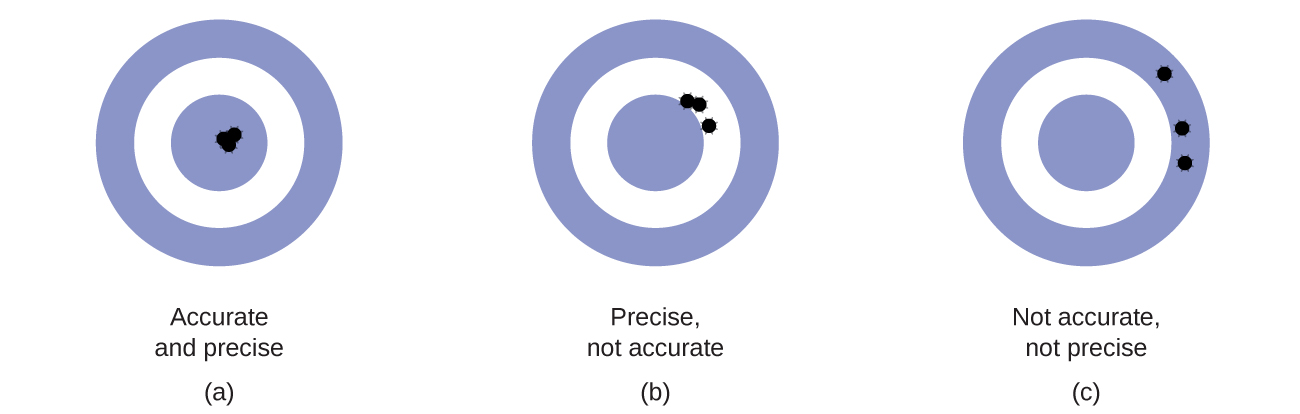
Suppose a quality control chemist at a pharmaceutical company is tasked with checking the accuracy and precision of three different machines that are meant to dispense 10 ounces (296 mL) of cough syrup into storage bottles. She proceeds to use each machine to fill five bottles and then carefully determines the actual volume dispensed, obtaining the results tabulated in [link] .
| Volume (mL) of Cough Medicine Delivered by 10-oz (296 mL) Dispensers | ||
|---|---|---|
| Dispenser #1 | Dispenser #2 | Dispenser #3 |
| 283.3 | 298.3 | 296.1 |
| 284.1 | 294.2 | 295.9 |
| 283.9 | 296.0 | 296.1 |
| 284.0 | 297.8 | 296.0 |
| 284.1 | 293.9 | 296.1 |
Considering these results, she will report that dispenser #1 is precise (values all close to one another, within a few tenths of a milliliter) but not accurate (none of the values are close to the target value of 296 mL, each being more than 10 mL too low). Results for dispenser #2 represent improved accuracy (each volume is less than 3 mL away from 296 mL) but worse precision (volumes vary by more than 4 mL). Finally, she can report that dispenser #3 is working well, dispensing cough syrup both accurately (all volumes within 0.1 mL of the target volume) and precisely (volumes differing from each other by no more than 0.2 mL).
Quantities can be exact or measured. Measured quantities have an associated uncertainty that is represented by the number of significant figures in the measurement. The uncertainty of a calculated value depends on the uncertainties in the values used in the calculation and is reflected in how the value is rounded. Measured values can be accurate (close to the true value) and/or precise (showing little variation when measured repeatedly).
Express each of the following numbers in scientific notation with correct significant figures:
(a) 711.0
(b) 0.239
(c) 90743
(d) 134.2
(e) 0.05499
(f) 10000.0
(g) 0.000000738592
Express each of the following numbers in exponential notation with correct significant figures:
(a) 704
(b) 0.03344
(c) 547.9
(d) 22086
(e) 1000.00
(f) 0.0000000651
(g) 0.007157
(a) 7.04 10 2 ; (b) 3.344 10 −2 ; (c) 5.479 10 2 ; (d) 2.2086 10 4 ; (e) 1.00000 10 3 ; (f) 6.51 10 −8 ; (g) 7.157 10 −3
Indicate whether each of the following can be determined exactly or must be measured with some degree of uncertainty:
(a) the number of eggs in a basket
(b) the mass of a dozen eggs
(c) the number of gallons of gasoline necessary to fill an automobile gas tank
(d) the number of cm in 2 m
(e) the mass of a textbook
(f) the time required to drive from San Francisco to Kansas City at an average speed of 53 mi/h
Indicate whether each of the following can be determined exactly or must be measured with some degree of uncertainty:
(a) the number of seconds in an hour
(b) the number of pages in this book
(c) the number of grams in your weight
(d) the number of grams in 3 kilograms
(e) the volume of water you drink in one day
(f) the distance from San Francisco to Kansas City
(a) exact; (b) exact; (c) uncertain; (d) exact; (e) uncertain; (f) uncertain
How many significant figures are contained in each of the following measurements?
(a) 38.7 g
(b) 2 10 18 m
(c) 3,486,002 kg
(d) 9.74150 10 −4 J
(e) 0.0613 cm 3
(f) 17.0 kg
(g) 0.01400 g/mL
How many significant figures are contained in each of the following measurements?
(a) 53 cm
(b) 2.05 10 8 m
(c) 86,002 J
(d) 9.740 10 4 m/s
(e) 10.0613 m 3
(f) 0.17 g/mL
(g) 0.88400 s
(a) two; (b) three; (c) five; (d) four; (e) six; (f) two; (g) five
The following quantities were reported on the labels of commercial products. Determine the number of significant figures in each.
(a) 0.0055 g active ingredients
(b) 12 tablets
(c) 3% hydrogen peroxide
(d) 5.5 ounces
(e) 473 mL
(f) 1.75% bismuth
(g) 0.001% phosphoric acid
(h) 99.80% inert ingredients
Round off each of the following numbers to two significant figures:
(a) 0.436
(b) 9.000
(c) 27.2
(d) 135
(e) 1.497 10 −3
(f) 0.445
(a) 0.44; (b) 9.0; (c) 27; (d) 140; (e) 1.5 10 −3 ; (f) 0.44
Round off each of the following numbers to two significant figures:
(a) 517
(b) 86.3
(c) 6.382 10 3
(d) 5.0008
(e) 22.497
(f) 0.885
Perform the following calculations and report each answer with the correct number of significant figures.
(a) 628 342
(b) (5.63 10 2 ) (7.4 10 3 )
(c)
(d) 8119 0.000023
(e) 14.98 + 27,340 + 84.7593
(f) 42.7 + 0.259
(a) 2.15 10 5 ; (b) 4.2 10 6 ; (c) 2.08; (d) 0.19; (e) 27,440; (f) 43.0
Perform the following calculations and report each answer with the correct number of significant figures.
(a) 62.8 34
(b) 0.147 + 0.0066 + 0.012
(c) 38 95 1.792
(d) 15 – 0.15 – 0.6155
(e)
(f) 140 + 7.68 + 0.014
(g) 28.7 – 0.0483
(h)
Consider the results of the archery contest shown in this figure.
(a) Which archer is most precise?
(b) Which archer is most accurate?
(c) Who is both least precise and least accurate?

(a) Archer X; (b) Archer W; (c) Archer Y
Classify the following sets of measurements as accurate, precise, both, or neither.
(a) Checking for consistency in the weight of chocolate chip cookies: 17.27 g, 13.05 g, 19.46 g, 16.92 g
(b) Testing the volume of a batch of 25-mL pipettes: 27.02 mL, 26.99 mL, 26.97 mL, 27.01 mL
(c) Determining the purity of gold: 99.9999%, 99.9998%, 99.9998%, 99.9999%

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?