| << Chapter < Page | Chapter >> Page > |
Electrolytes, such as sodium chloride, ionize in water, meaning that they dissociate into their component ions. In water, sodium chloride (NaCl), dissociates into the sodium ion (Na + ) and the chloride ion (Cl – ). The most important ions, whose concentrations are very closely regulated in body fluids, are the cations sodium (Na + ), potassium (K + ), calcium (Ca +2 ), magnesium (Mg +2 ), and the anions chloride (Cl - ), carbonate (CO 3 -2 ), bicarbonate (HCO 3 - ), and phosphate(PO 3 - ). Electrolytes are lost from the body during urination and perspiration. For this reason, athletes are encouraged to replace electrolytes and fluids during periods of increased activity and perspiration.
Osmotic pressure is influenced by the concentration of solutes in a solution. It is directly proportional to the number of solute atoms or molecules and not dependent on the size of the solute molecules. Because electrolytes dissociate into their component ions, they, in essence, add more solute particles into the solution and have a greater effect on osmotic pressure, per mass than compounds that do not dissociate in water, such as glucose.
Water can pass through membranes by passive diffusion. If electrolyte ions could passively diffuse across membranes, it would be impossible to maintain specific concentrations of ions in each fluid compartment therefore they require special mechanisms to cross the semi-permeable membranes in the body. This movement can be accomplished by facilitated diffusion and active transport. Facilitated diffusion requires protein-based channels for moving the solute. Active transport requires energy in the form of ATP conversion, carrier proteins, or pumps in order to move ions against the concentration gradient.
In order to calculate osmotic pressure, it is necessary to understand how solute concentrations are measured. The unit for measuring solutes is the mole . One mole is defined as the gram molecular weight of the solute. For example, the molecular weight of sodium chloride is 58.44. Thus, one mole of sodium chloride weighs 58.44 grams. The molarity of a solution is the number of moles of solute per liter of solution. The molality of a solution is the number of moles of solute per kilogram of solvent. If the solvent is water, one kilogram of water is equal to one liter of water. While molarity and molality are used to express the concentration of solutions, electrolyte concentrations are usually expressed in terms of milliequivalents per liter (mEq/L): the mEq/L is equal to the ion concentration (in millimoles) multiplied by the number of electrical charges on the ion. The unit of milliequivalent takes into consideration the ions present in the solution (since electrolytes form ions in aqueous solutions) and the charge on the ions.
Thus, for ions that have a charge of one, one milliequivalent is equal to one millimole. For ions that have a charge of two (like calcium), one milliequivalent is equal to 0.5 millimoles. Another unit for the expression of electrolyte concentration is the milliosmole (mOsm), which is the number of milliequivalents of solute per kilogram of solvent. Body fluids are usually maintained within the range of 280 to 300 mOsm.
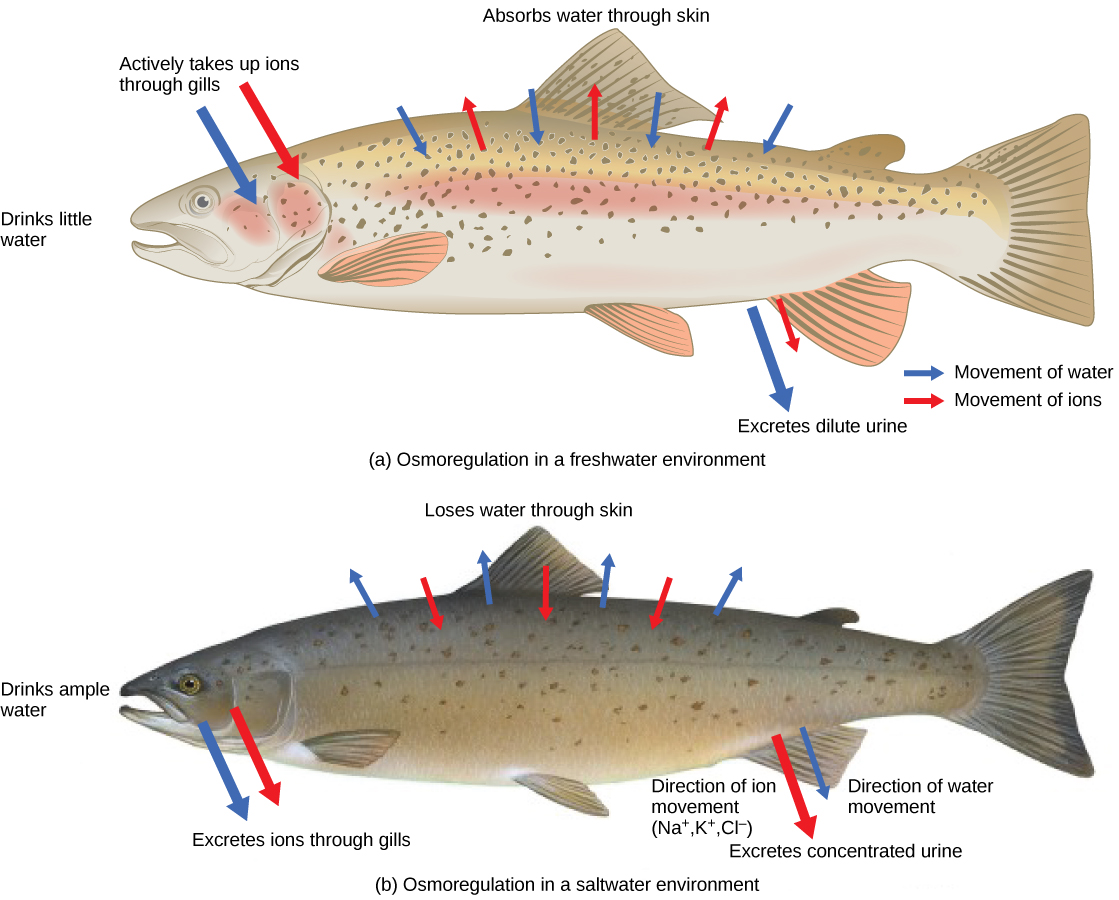
Persons lost at sea without any fresh water to drink are at risk of severe dehydration because the human body cannot adapt to drinking seawater, which is hypertonic in comparison to body fluids. Organisms such as goldfish that can tolerate only a relatively narrow range of salinity are referred to as stenohaline. About 90 percent of all bony fish are restricted to either freshwater or seawater. They are incapable of osmotic regulation in the opposite environment. It is possible, however, for a few fishes like salmon to spend part of their life in fresh water and part in sea water. Organisms like the salmon and molly that can tolerate a relatively wide range of salinity are referred to as euryhaline organisms. This is possible because some fish have evolved osmoregulatory mechanisms to survive in all kinds of aquatic environments. When they live in fresh water, their bodies tend to take up water because the environment is relatively hypotonic, as illustrated in [link] a . In such hypotonic environments, these fish do not drink much water. Instead, they pass a lot of very dilute urine, and they achieve electrolyte balance by active transport of salts through the gills. When they move to a hypertonic marine environment, these fish start drinking sea water; they excrete the excess salts through their gills and their urine, as illustrated in [link] b . Most marine invertebrates, on the other hand, may be isotonic with sea water ( osmoconformers ). Their body fluid concentrations conform to changes in seawater concentration. Cartilaginous fishes’ salt composition of the blood is similar to bony fishes; however, the blood of sharks contains the organic compounds urea and trimethylamine oxide (TMAO). This does not mean that their electrolyte composition is similar to that of sea water. They achieve isotonicity with the sea by storing large concentrations of urea. These animals that secrete urea are called ureotelic animals. TMAO stabilizes proteins in the presence of high urea levels, preventing the disruption of peptide bonds that would occur in other animals exposed to similar levels of urea. Sharks are cartilaginous fish with a rectal gland to secrete salt and assist in osmoregulation.
Dialysis technicians typically work in hospitals and clinics. While some roles in this field include equipment development and maintenance, most dialysis technicians work in direct patient care. Their on-the-job duties, which typically occur under the direct supervision of a registered nurse, focus on providing dialysis treatments. This can include reviewing patient history and current condition, assessing and responding to patient needs before and during treatment, and monitoring the dialysis process. Treatment may include taking and reporting a patient’s vital signs and preparing solutions and equipment to ensure accurate and sterile procedures.
Solute concentrations across a semi-permeable membranes influence the movement of water and solutes across the membrane. It is the number of solute molecules and not the molecular size that is important in osmosis. Osmoregulation and osmotic balance are important bodily functions, resulting in water and salt balance. Not all solutes can pass through a semi-permeable membrane. Osmosis is the movement of water across the membrane. Osmosis occurs to equalize the number of solute molecules across a semi-permeable membrane by the movement of water to the side of higher solute concentration. Facilitated diffusion utilizes protein channels to move solute molecules from areas of higher to lower concentration while active transport mechanisms are required to move solutes against concentration gradients. Osmolarity is measured in units of milliequivalents or milliosmoles, both of which take into consideration the number of solute particles and the charge on them. Fish that live in fresh water or saltwater adapt by being osmoregulators or osmoconformers.

Notification Switch
Would you like to follow the 'Biology' conversation and receive update notifications?