| << Chapter < Page | Chapter >> Page > |
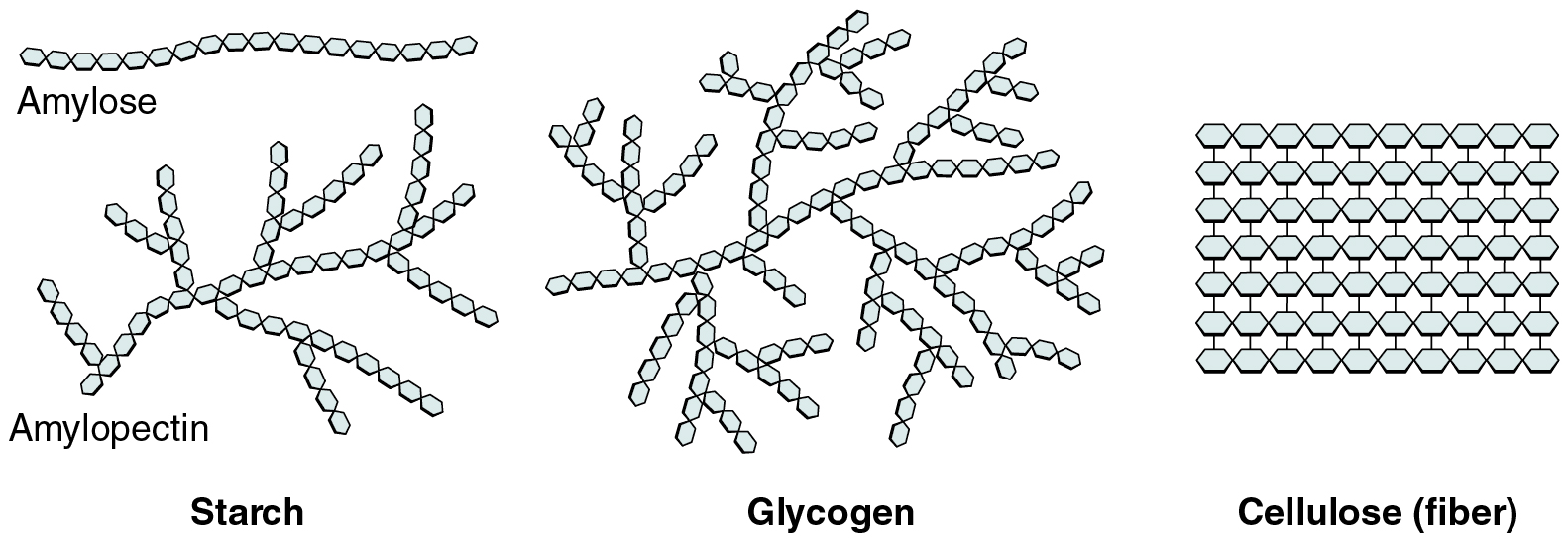
Polysaccharides can contain a few to a thousand or more monosaccharides. Three are important to the body ( [link] ):
The body obtains carbohydrates from plant-based foods. Grains, fruits, and legumes and other vegetables provide most of the carbohydrate in the human diet, although lactose is found in dairy products.
Although most body cells can break down other organic compounds for fuel, all body cells can use glucose. Moreover, nerve cells (neurons) in the brain, spinal cord, and through the peripheral nervous system, as well as red blood cells, can use only glucose for fuel. In the breakdown of glucose for energy, molecules of adenosine triphosphate, better known as ATP, are produced. Adenosine triphosphate (ATP) is composed of a ribose sugar, an adenine base, and three phosphate groups. ATP releases free energy when its phosphate bonds are broken, and thus supplies ready energy to the cell. More ATP is produced in the presence of oxygen (O 2 ) than in pathways that do not use oxygen. The overall reaction for the conversion of the energy in glucose to energy stored in ATP can be written:
In addition to being a critical fuel source, carbohydrates are present in very small amounts in cells’ structure. For instance, some carbohydrate molecules bind with proteins to produce glycoproteins, and others combine with lipids to produce glycolipids, both of which are found in the membrane that encloses the contents of body cells.
A lipid is one of a highly diverse group of compounds made up mostly of hydrocarbons. The few oxygen atoms they contain are often at the periphery of the molecule. Their nonpolar hydrocarbons make all lipids hydrophobic. In water, lipids do not form a true solution, but they may form an emulsion, which is the term for a mixture of solutions that do not mix well.
A triglyceride is one of the most common dietary lipid groups, and the type found most abundantly in body tissues. This compound, which is commonly referred to as a fat, is formed from the synthesis of two types of molecules ( [link] ):

Notification Switch
Would you like to follow the 'Anatomy & Physiology' conversation and receive update notifications?