| << Chapter < Page | Chapter >> Page > |
The products of nucleic acid digestion—pentose sugars, nitrogenous bases, and phosphate ions—are transported by carriers across the villus epithelium via active transport. These products then enter the bloodstream.
The electrolytes absorbed by the small intestine are from both GI secretions and ingested foods. Since electrolytes dissociate into ions in water, most are absorbed via active transport throughout the entire small intestine. During absorption, co-transport mechanisms result in the accumulation of sodium ions inside the cells, whereas anti-port mechanisms reduce the potassium ion concentration inside the cells. To restore the sodium-potassium gradient across the cell membrane, a sodium-potassium pump requiring ATP pumps sodium out and potassium in.
In general, all minerals that enter the intestine are absorbed, whether you need them or not. Iron and calcium are exceptions; they are absorbed in the duodenum in amounts that meet the body’s current requirements, as follows:
Iron —The ionic iron needed for the production of hemoglobin is absorbed into mucosal cells via active transport. Once inside mucosal cells, ionic iron binds to the protein ferritin, creating iron-ferritin complexes that store iron until needed. When the body has enough iron, most of the stored iron is lost when worn-out epithelial cells slough off. When the body needs iron because, for example, it is lost during acute or chronic bleeding, there is increased uptake of iron from the intestine and accelerated release of iron into the bloodstream. Since women experience significant iron loss during menstruation, they have around four times as many iron transport proteins in their intestinal epithelial cells as do men.
Calcium —Blood levels of ionic calcium determine the absorption of dietary calcium. When blood levels of ionic calcium drop, parathyroid hormone (PTH) secreted by the parathyroid glands stimulates the release of calcium ions from bone matrices and increases the reabsorption of calcium by the kidneys. PTH also upregulates the activation of vitamin D in the kidney, which then facilitates intestinal calcium ion absorption.
The small intestine absorbs the vitamins that occur naturally in food and supplements. Fat-soluble vitamins (A, D, E, and K) are absorbed along with dietary lipids in micelles via simple diffusion. This is why you are advised to eat some fatty foods when you take fat-soluble vitamin supplements. Most water-soluble vitamins (including most B vitamins and vitamin C) also are absorbed by simple diffusion. An exception is vitamin B 12 , which is a very large molecule. Intrinsic factor secreted in the stomach binds to vitamin B 12 , preventing its digestion and creating a complex that binds to mucosal receptors in the terminal ileum, where it is taken up by endocytosis.
Each day, about nine liters of fluid enter the small intestine. About 2.3 liters are ingested in foods and beverages, and the rest is from GI secretions. About 90 percent of this water is absorbed in the small intestine. Water absorption is driven by the concentration gradient of the water: The concentration of water is higher in chyme than it is in epithelial cells. Thus, water moves down its concentration gradient from the chyme into cells. As noted earlier, much of the remaining water is then absorbed in the colon.
The small intestine is the site of most chemical digestion and almost all absorption. Chemical digestion breaks large food molecules down into their chemical building blocks, which can then be absorbed through the intestinal wall and into the general circulation. Intestinal brush border enzymes and pancreatic enzymes are responsible for the majority of chemical digestion. The breakdown of fat also requires bile.
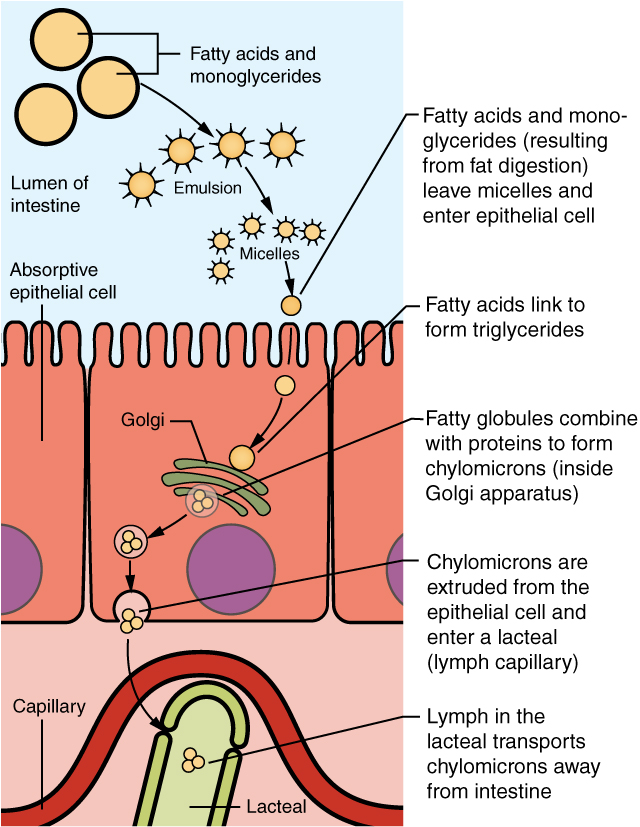
Most nutrients are absorbed by transport mechanisms at the apical surface of enterocytes. Exceptions include lipids, fat-soluble vitamins, and most water-soluble vitamins. With the help of bile salts and lecithin, the dietary fats are emulsified to form micelles, which can carry the fat particles to the surface of the enterocytes. There, the micelles release their fats to diffuse across the cell membrane. The fats are then reassembled into triglycerides and mixed with other lipids and proteins into chylomicrons that can pass into lacteals. Other absorbed monomers travel from blood capillaries in the villus to the hepatic portal vein and then to the liver.

Notification Switch
Would you like to follow the 'Anatomy & Physiology' conversation and receive update notifications?